Organic Chemistry Milestones
For timelines on the development of the periodic table and some of the history of chemistry, please visit: http://murov.info/timelines.htm .

CHRONOLOGY
| year |
contributor |
image |
Location |
contribution |
| 8000 BC |
|
|
|
|
| 340 BC | Aristotle |
 |
Greece | The concept of vitalism (that compounds derived from plants and animals contain a property derivable only from the living) seems to have been derived from the philosophy of Aristotle. |
| 1250 | Taddeo Alderotti |
 |
Italy | Alderotti was an alchemist who is given credit for the development of fractional distillation. |
| 1540 | Valerius Cordus |
 |
Germany | Cordus (a student of Paracelsus) synthesized diethyl ether by the action of sulfuric acid on ethanol. Paracelsus noticed ether's anesthetic properties (about 1540). William Morton (1846) and shortly thereafter, Crawford Long demonstrated ether's use as a surgical anesthetic. |
| 1746 | John Roebuck |
 |
England | Sulfuric acid is used to catalyze many organic reactions as noted with the synthesis of diethyl ether from ethanol. However, Roebuck developed the first industrial scale synthesis of sulfuric acid. |
| 1770 | Torbern O. Bergman |
 |
Sweden | Bergman was the first to distinguish between organic and inorganic chemistry with organic being associated with chemicals derived from living things and containing a "property" called vitalism. |
| 1806 |
N-L. Vauquelin, P-J. Robiquet |
  |
France | Vauquelin and his student Robiquet isolated the first known amino acid. It was not until a century later that Emil Fischer and Hofmeister independently showed that proteins are polymers made from amino acids. |
|
1808, 1812 |
E-L. Malus, J-B. Biot |
 |
France | Malus and 4 years later, Biot developed the polarimeter. Biot discovered in 1815 that solutions of tartaric acid can be optically active. |
|
1920 - 1932 |
H. Wieland, A. Windaus |
  |
In 1758, Francois Poulletier de La Salle isolated cholesterol from gallstones and Michel Eugene Chevreu extended the research in 1815 and called the compound cholesterol. However, it was more than a century later when Heinrich Wieland & Adolf Windaus determined the structure of cholesterol and bile acids. | |
| 1828 | Friedrich Wöhler |
 |
Germany | By synthesizing urea from the "inorganic" ammonium cyanate, Wöhler disproved the vitalism concept (compounds derived from a living plant or animal contained a special characteristic called a vital force) that led to a redfining of organic chemistry from the chemistry of compounds derived from the living to compounds containing carbon and hydrogen and later to compounds of carbon. His experiment was one of the first demonstrations of isomerism. He also was one of the first to initiate the concept of functional groups. |
| 1820's | Jons Jacob Berzelius |
 |
Sweden | Berzelius coined the terms isomerism, catalyst and protein and reportedly noticed that lactic acid has two optical isomers. |
| 1820's | J. L. Gay Lussac, J. F. von Liebig |
  |
Lussac developed the method of combustion analysis and von Liebig made the method practical for the determination of the percent of carbon in organic compounds. Combustion analysis opened up the field of organic chemistry. von Liebig and Wöhler also initiated the concept of functional groups. | |
| 1849 | Louis Pasteur |
 |
France | Although best known for his work on vaccination, microbial fermentation and pasteurization, Pasteur made a very significant contribution to organic chemistry when he extentded Biot's work and achieved the first resolution of a racemic mixture of tartaric acid and laid the foundation of what we now call stereochemistry. |
| 1856 | William Henry Perkin |
 |
England | Perkin, when attempting to synthesize quinine, serendipitously synthesized mauveine and had a successful career in dye synthesis. |
| 1862 | Emil Erlenmeyer |
 |
Germany | While known best for the Erlenmeyer flask, Erlenmeyer performed many original syntheses, developed the Erlenmeyer rule and very importantly was the first to suggest the possibility of double and triple bonds between two carbons. |
| 1865 | August Kekulé |
 |
Germany | Kekulé was one of the first to draw structures of organic compounds using lines for bonds and is most famous for his proposed Kekulé structure of benzene. |
| 1869 | John Wesley Hyatt |
 |
The origin of polymer science has been attributed to many different people. John Wesley Hyatt has been given credit for preparing the first synthetic polymer. In 1839, Johann Eduard Simon accidentally prepared polystyrene. In 1909, Leo Baekeland synthesized bakellite. Hermann Staudinger in 1920 was the first to properly characterize polymers as macromolecules. | |
| 1870 | Vladimir V. Markovnikov |
 |
Russia | Markonikov is best known for his useful Markonikov's rule stating that in an addition of H-X to an alkene, the nucleophilic X- adds to the carbon atom with fewer hydrogen atoms, while the proton adds to the carbon atom with more hydrogen atoms bonded to it. |
| 1871 | Ernst Abbe |
 |
Germany | Abbe developed many optical tools including the refractometer. The Abbe refractometer enables precise measurements of refractive index which can help identify previously known compounds. |
| 1874 | J. H. van 't Hoff, Joseph A. Le Bel |
  |
Netherlands, France | van 't Hoff and Le Bel developed the concept of the arrangement of atoms in space (3d molecules) including the tetrahedral arrangement of atoms bonded to a tetravalent carbon and the relationship to optical activity. |
| 1875 | Alexander Zaitsev |
 |
Russia | In an elimination reaction, the more highly substituted alkene (thermodynamically most stable) is the more likely product of an elimination reaction according to Zaitsev's rule. The rule is very accurate for E1 reactions but there are many exceptions for E2 reactions. |
| 1877 | Charles Friedel, James Crafts |
  |
France | Friedel and Crafts developed the Friedel-Crafts reaction involving alkylation and acylation reactions of aromatic rings, another type of valuable carbon-carbon bond forming reactions. |
| 1880 | Friedrich Beilstein |
 |
Russia, Germany | Beilstein systematically produced a listing of the properties of organic compounds in a handbook of organic chemistry later to be known as the Beilstein database. |
| 1881 | Ludwig Claisen |
 |
Germany | Claisen discovered many important reactions including the Claisen condensation and the Claisen rearrangement and designed the Claisen flask. |
| 1883 | Adolf von Baeyer |
 |
Germany | von Baeyer synthesized and determined the structure of indigo and other dyes. He also developed a nomenclature system for cyclic systems and synthesized the first baribiturate. |
| 1888, 1989 |
Friedrich Reinitzer, Otto Lehman |
  |
Austria, Germany | The year after Reinitzer observed the first liquid crystal, cholesterol benzoate, Otto Lehman published a paper on the properties of liquid crystal. In 1922 Geoges Friedel classified liquid crystals into three categories. For a timeline: see: http://www.iop.org/cs/page_43664.html |
| 1891 | Vladimir Grigoryevich Shukhov |
 |
Russia | Shukov devleoped thermal cracking. Cracking serves as the method for providing feedstock for the synthesis of many organic chemicals. William Burton is usually given credit for the first commercial application of cracking. With many advantages over thermal cracking, catalytic cracking was developed by Eugene Jules Houdry in 1937. |
| 1892 | Emil Fischer |
 |
Germany | Fischer was responsible for many major discoveries in organic chemistry. He elucidated the structures of sugars, developed the Fischer projection system for tetrahedral carbons, developed Fischer esterification, contributed to the elucidation of protein structure including development of the lock and key model for the behavior of peptide catalysts. |
| 1895 | Paul Walden |
 |
Russia, Latvia, Germany | Walden made significant contributions to the study of stereochemistry including the concept of the Walden inversion. |
| 1900 | F. A. Victor Grignard |
 |
France | The discovery that magnesium reacts with organohalides to form Grignard reagents that subsequently react with carbonyls (and other compounds) to form carbon - carbon bonds enabled the synthesis of many compounds. Care must be taken when running this reaction to exclude water. |
| 1900 | Moses Gomberg |
 |
U.S. | Gomberg, when trying to make hexaphenylethane, recognized that he was the first to prepare a stable free radical, triphenylmethyl. |
| 1903 |
Mikhail
Tswett (Tsvet) |
 |
Russia | Tswett while performing research on plant pigments developed the technique of column chromatography. HPLC was a direct develpment from this work and other types of chromatograhy including gas and thin layer were derived from the innovative discovery of Tswett. |
| 1903 | William W. Coblentz |
 |
U.S. | Sir William Herschel discovered infrared spectroscopy in 1800 but it was not until 1903 that Coblentz did the ground breaking work that eventually enabled ir to become a valuable and routine technique for functional group determination and some structural analysis of organic compounds. |
| ~1910 | Giacomo Luigi Ciamician |
 |
Ciamician was a pioneer in the field of organic photochemistry. 50 years after he initiated the use of light to energized organic chemical reactions, the field was greatly expanded by several researchers including George Hammond and his proteges and Georger Porter. | |
| 1916 | Gilbert Lewis |
 |
U.S. | Lewis developed the concepts of electron pairs, bonding, Lewis structures and Lewis acids (1923) for atoms and molecules. He, expanded work on the valence concepts of Richard Abegg. |
| 1920 |
Wendell Latimer, Worth Rodebush |
  |
T. S. Moore and T. F. Winmill first applied the concept of the hydrogen bond in 1912 but Latimer and Rodebush were the first to describe hydrogen bonding. Hydrogen bonding is responsible for many several extremely important properties including the boiling poin of water and the structures of DNA and proteins. | |
| 1922 | Francis Aston |
 |
England | Aston invented the mass spectrometer, an extremely powerful method for determining the structure and identification of organic compounds |
| 1922... | Christopher Ingold |
 |
England | Ingold opened up the field of mechanistic studies of organic reactions while developing descriptions and terminology for nucleophilic and electrophilic substitution reactions. |
| 1923 | Johannes N. Brønsted, Thomas M. Lowry |
  |
Denmark, England | Brønsted and Lowry indepently proposed that a Brønsted acid should be defined as a proton donor and a base as a proton acceptor. |
| 1928 |
Otto P. H.
Diels Kurt Alder |
 |
Germany | The Diels-Alder reaction is a [4+2] cycloaddition) between a conjugated diene and a substituted alkene to form a substituted cyclohexene. Reactions that form carbon - carbon bonds are extremely useful. |
| 1929 | Kathleen Lonsdale |
 |
Ireland | Lonsdale proved using x-ray chrystallography that benzene is flat and all bonds are the same length (not alternating single and double bonds). |
| 1930, 1934 | Wallace Carothers |
 |
U.S. | Carothers and Elmer Bolton discovered neoprene and Crothers and Julian Hill discovered nylon and the useful synthetic techniques for producing these polymers. |
| 1931 | Erich Hückel |
 |
Germany | Hückel developed Hückel's rule that states that a planar ring with 4n + 2 p electrons should have a special stabilizing form of resonance called aromaticity. |
| 1935 | Louis Hammett |
 |
U.S. | The Hammett equation (a free energy relationship) enabled the calculation of a plethora of reaction rates and equilibrium constants from a table of substituent constants and reaction properties. The constants also enable a comparision of the magnitudes of substituent effects. |
| 1938 |
Isidor I. Rabi, Felix Bloch, Edward Purcell |
 |
U.S., Switzerland, U.S. | Rabi, Block and Purcell developed the extremely powerful technique of nuclear magnetic resonance that is currently used routinely for the determination of the structures of organic compounds. The concept was eventually developed into the very valuable technique, MRI, for imaging portions of the human body. Martin Packard was the first to record an nmr of an organic compound, James Arnold and later James Shoolery developed the instrumentation and John Roberts demonstrated the applications in organic chemistry. |
| 1939 | N. A. Izmailov, M.S. Shraiber |
 |
Russia | Ismailov and Shraiber developed the technique of thin layer chromatography. |
|
1940 - 1979 |
Robert Woodward |
 |
U.S. | Woodward and his research group synthesized many natural products including quinine, cholesterol, cortisone, strychnine, lysergic acid, reserpine, chlorophyll, cephalosporin and colchicine and Vitamin B12. Along the way, he developed many new synthetic methods and the Woodward - Hoffmann rules (see Fukii - 1952 - and Hoffmann - 1965) |
| 1941-1952 |
Archer Martin, Richard Synge, Anthony James, Fritz Prior, Erika Cremer |
 |
England, Austria | Martin and Synge received Nobel prize in 1952 for development of partition chromatography with contributions to gas chromatography by James, Prior and Cremer. |
| 1947 | J. Bigeleisen, Maria Goeppert Mayer |
  |
U.S. | Bigeleisen and Mayer published the first paper on the kinetic isoptope effect on reaction rates and equilbria. Use of isotope substitution provides considerable mechanistic insight into organic reactions. |
| 1949 | Peter Fellgett, Pierre Jacquinot |
  |
England, France | The application of fourier transform methodolgy to ir was made possible by the combined contributions of Fellgett and Jacquinot. |
| 1950 | Derek Barton |
 |
England | Barton connected the conformation of rings including steroids to reactivity. |
| 1950 | Roland Gohlke, F. McLafferty |
  |
U.S. | Gohlke and McLafferty used a mass spectrometer as the detector for a gas chromatograph resulting in the the very powerful and fast technique gcms that is capable of separating and identifying components of mixtures and providing extensive structural information simultaneously. |
| 1950 | Lyman C. Craig |
 |
Craig invented the rotary evaporator and other useful types of apparatus. In 1957, Büchi commercialized the rotary evaporator. | |
| 1952 | Kenichi Fukui |
 |
Japan | Fukui developed a method involving the use of frontier molecular orbitals of explaining and predicting the course of synchronous organic reactions. The method was later made more practical with the development of the Woodward - Hoffmann rules for pericyclic reactions. |
| 1954 | Vladimir Prelog |
 |
Bosnia | Prelog performed ground breaking work connecting reactivity to organic structure especially with regard to steroisomerism. Along with Cahn and Ingold, he developed a system for distinguishing and naming enantiomers. |
| 1954 | Georg Wittig |
 |
Germany | The Wittig reaction for synthesizing alkenes from carbonyls and ylides like other reactions that form carbon - carbon bonds was a very valuable addition to the repertoire of synthetic organic chemistry. |
| 1955 | George Hammond |
 |
U.S. | The Hammond postulate suggests that it is possible to obtain information about transition states by examining the structure of the nearest stable species. His research group contributed to the opening up of the field of organic photochemistry. |
| 1957-- | Elias J. Corey |
 |
U.S. | Corey developed many original organic synthetic techniques and pioneered the use of retrosynthetic approaches. Corey was also one of the first to use computerized approaches for organic synthesis. |
| 1959 | Herbert C. Brown |
 |
U.S. | Brown developed many organoboron reactions and the use of borohydrides for reduction. He is most famous for the anti-Markovnikov hydroboration reaction. |
| 1965 | Roald Hoffmann |
 |
U.S. | Woodward and Hoffmann developed a series of rules based on molecular orbitals that explain and predict the stereochemistry of pericyclic reactions. Also see Woodward (1940) and Fukui (1952). |
| 1966 |
Richard Ernst, Weston Anderson |
 |
Switzerland, U.S. | Ernst and Anderson applied the use of fourier transform technology to nmr vastly improving its sensitivity. |
| 1985 |
Harold Kroto, R. Smalley, Robert Curl |
 |
U.S., England, U.S. | Kroto, Smalley and Curl dicovered methods for synthesizing and characterizing buckminister fullerene (also called bucky balls and fullerene). This work undoutedly helped with the discovery of nanotubes and graphene. |
| 1986 | K. C. Westaway, R. N. Gedye |
 |
England | Percy Spencer discovered the potential for microwave heating in 1946 but it was not until 1986 that microwave ovens were added to the repertoire of heating techniques available to the organic chemist by the publications of Westaway and Gedye. |
| 1991 | Sumio Iijima |
 |
Japan | Iijima made synthesis of carbon nanotubes practical but his work followed on the shoulders of many others including Radushkevich, Lukyanovich, R. Bacon, and M. Endo. For a timeline of nanotube discoveries, see: https://en.wikipedia.org/wiki/Timeline_of_carbon_nanotubes |
| 2004 |
Andre Geim, K. Novoselov |
 |
England | Geim and Novoselov performed groundbreaking research about graphene and its two dimensional properties. |
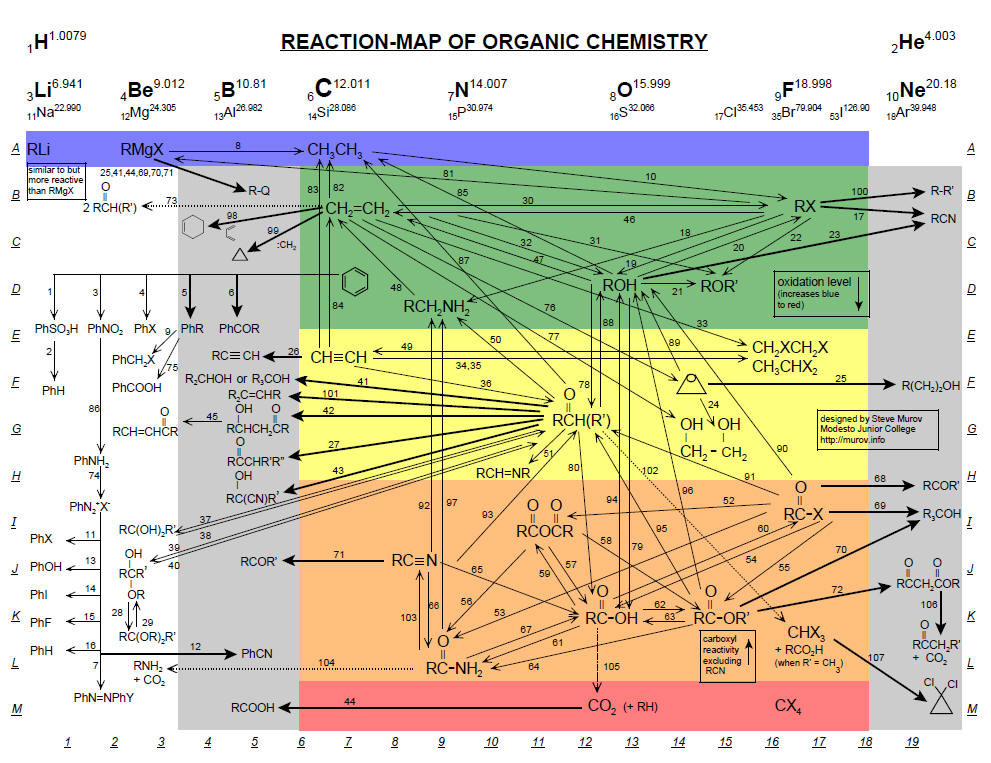
The reaction map and accompnaying keys are available using the link at: http://murov.info/Reaction-Map.htm
Other websites by this author are available at: http://murov.info/
Organic chemistry web sites by this author include:
Properties of organic solvents and hotlinks to organic chemical property sites: http://murov.info/orgsolvents.htm
Organic chemistry directory: http://murov.info/orgchem.htm
Online organic laboratory text: http://murov.info/orglab/orglab.htm
Organic chemistry exercises (also included in organic laboratory text): http://murov.info/orglab/exercises.htm
Web site originally posted on 6/10/18 by Steve Murov. http://murov.info
Selection of milestones has been made subjectively and
apologies in advance to those who think significant contributions have not been
included.
If you have a case for the addition of additional milestones,
your suggestions will be very much appreciated and considered for addition to
the list.
murovs@yosemite.edu